Peripheral arterial disease (PAD) is a severe and growing problem affecting the quality of life of over 200 million people worldwide.1
Endovascular treatment has been validated as first-line therapy for PAD. Several techniques and tools are available, supported by different levels and quantities of clinical evidence.2
Since their introduction in Europe in 2008, drug-coated balloons (DCBs) have increasingly been used for lower limb revascularisation. Following initial excellent results reported in the literature, many physicians started to use DCBs daily as a first-line therapy in the femoropopliteal region, supported by level 1 evidence.3,4 DCBs achieve high rates of primary patency (PP), compared with alternative therapies, with a limited use of provisional stents.
Clinical Studies
DCBs act on the smooth muscle cells reducing intimal hyperplasia and the risk of restenosis. In recent years many clinical studies have been performed comparing DCBs with conventional percutaneous transluminal angioplasty (PTA). All of these studies have shown superiority of DCBs in late lumen loss (LLL) and in PP. More than 13 DCBs are available on the market in Europe, but not all of them have been evaluated with robust clinical studies.
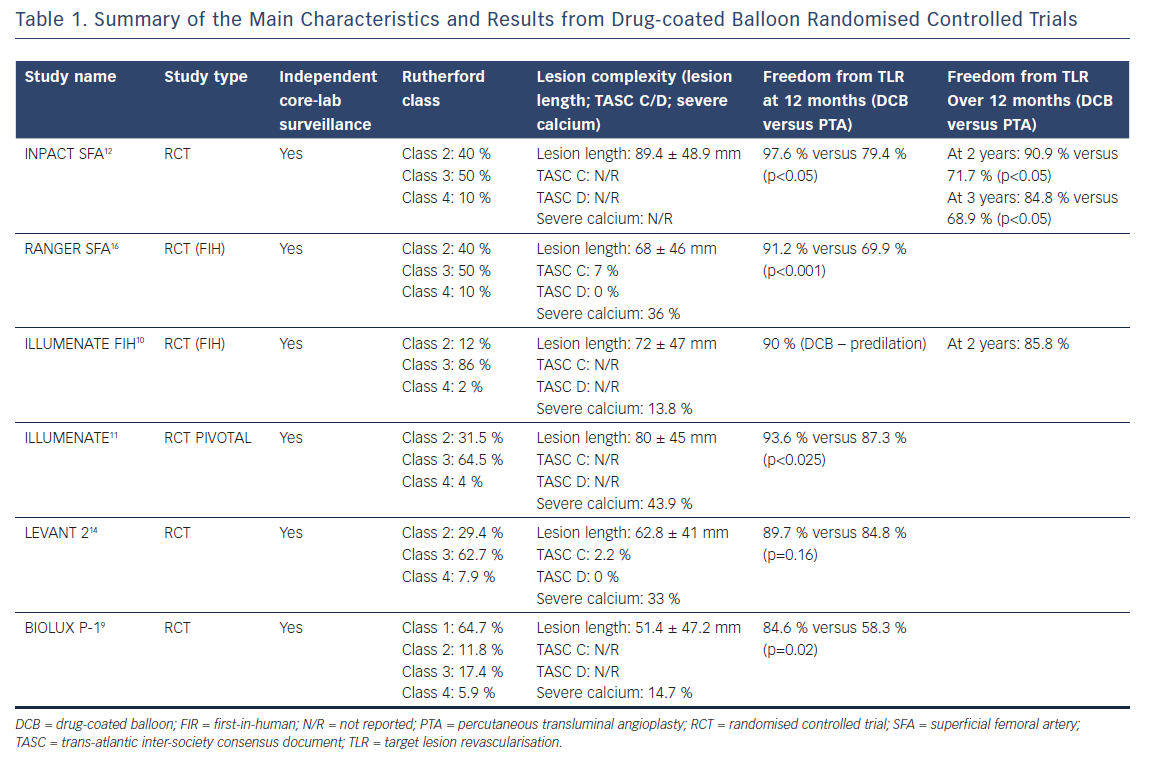
In the first milestone trial – Local Taxan With Short Time Contact for Reduction of Restenosis in Distal Arteries (THUNDER) – Tepe et al. reported a great advantage in the use of the prototype DCB over traditional angioplasty, in terms of LLL (0.7 ± 1.9 versus 1.5 ± 1.3 mm; p=0.5) and binary restenosis (BR) (17 % versus 54 %; p=0.04). The advantage of DCB has been maintained over more than 5 years.5,6
More recently many other studies have confirmed similar results, not only after 12-month follow-up, but also after 24 and 36 months.7–15 1-year PP was 89.5 % in the Pivotal Trial of a Novel Paclitaxel-Coated Percutaneous Angioplasty Balloon (ILLUMENATE),10,11 82.2 % in the Randomized Trial of IN.PACT Admiral® Drug Coated Balloon vs Standard PTA for the Treatment of SFA and Proximal Popliteal Arterial Disease (INPACT SFA),12 73.5 % in the Moxy Drug Coated Balloon versus Standard Balloon Angioplasty for the Treatment of Femoropopliteal Arteries (LEVANT 2) study14,15 and 86.4 % in the Comparison of the Ranger™ Paclitaxel-coated PTA Balloon Catheter and Uncoated PTA Balloons in Femoropopliteal Arteries (RANGER SFA) trial.16 After 2 years of follow-up, PP was 80.3 % for the Stellarex™ DCB, 78.9 % for the IN.PACT® Admiral® DCB (Medtronic) and 58.6 % for the Lutonix® DCB (Bard).
Initial analysis of these data underlines the concept of a prolonged effect of paclitaxel, confirmed by the difference in PP when compared with PTA after 2 years of follow-up. Three-year data are also available for the INPACT SFA trial, confirming the long-term efficacy of the DCB balloon versus PTA. A 3-year PP of 69.5 % (versus 45.1 % with PTA) and a clinically driven target lesion revascularisation (CD TLR) of 2.4 % at 1 year, 9.1 % at 2 years and 15.2 % at 3 years (versus a PTA CD TLR of 20.6 %, 28.3 % and 31.1 % at 1, 2 and 3 years, respectively; p<0.05) have been described.17 Table 1 summarises the main characteristics and outcomes of the most recent randomised controlled trials.
Real-world Data
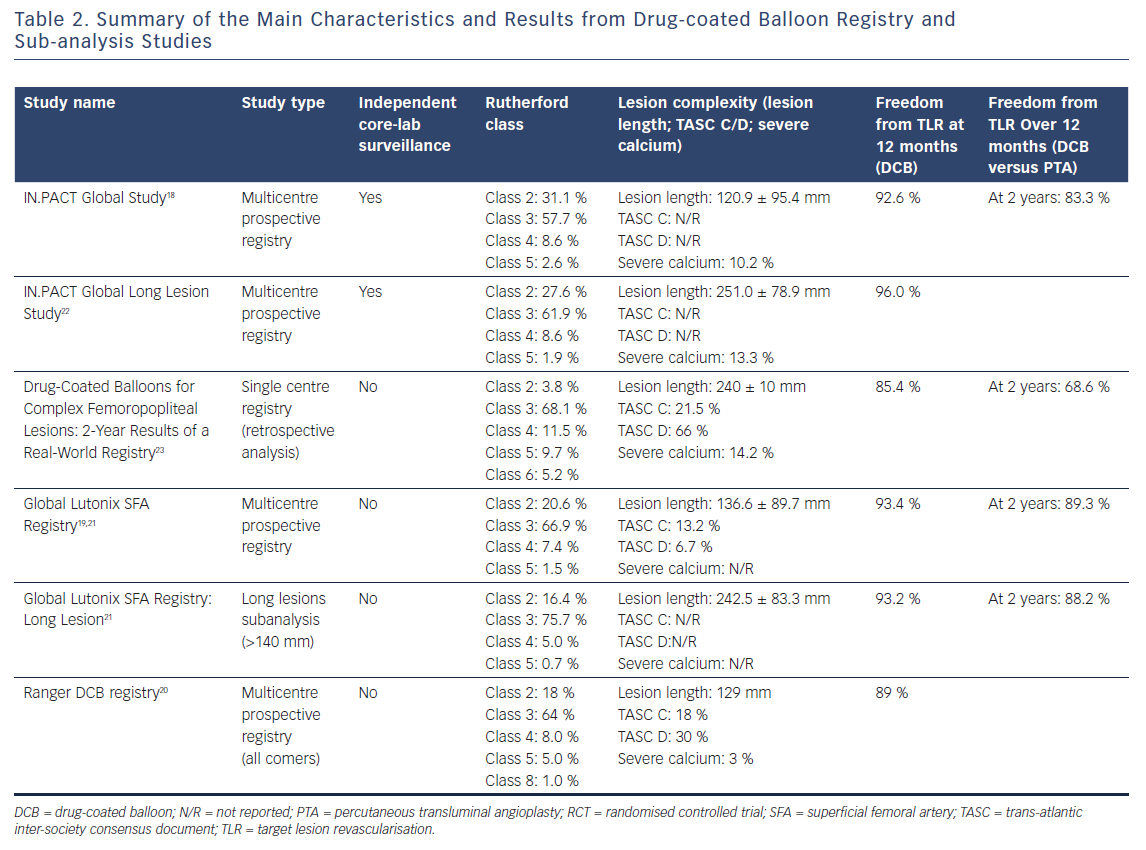
Data coming from global registries are interesting and represent more of a real-world situation. Three studies have been conducted: the IN.PACT Global Study with the IN.PACT Admiral DCB.18 The Global SFA (superficial femoral artery) Registry with the Lutonix DCB19 and Ranger DCB Registry with the Ranger DCB (Boston Scientific).20
In the IN.PACT Global Study,18 1,406 patients with Rutherford class from two to four were enrolled. Mean lesion length was 12.09 cm with 35 % total occlusion and 18 % in-stent restenosis (ISR). Freedom from CD TLR (primary endpoint) was 92.6 % at 360 days (evaluated by Kaplan-Meier analysis and adjudicated by an independent clinical event committee).
In the Global SFA Registry, 691 patients with Rutherford class from two to six were enrolled. Mean lesion length was 101.2 mm with 31.2 % total occlusion. Freedom from TLR at 12 months was 93.6 % for the whole group of patients and 94.7 % for patients with total occlusion. Again, in this study results were evaluated with Kaplan–Meier analysis. Interim results at 24 months showed a freedom from TLR of 89.2 %.19
In the Ranger DCB All-comers Registry, patients with Rutherford class from one to six were enrolled. Mean lesion length was 129.0 mm and severe calcification was present in only 3 % of lesions. Freedom from TLR was 89.2 %, with significant improvement of the Rutherford class at 12 months.20
A study of the results of the Lutonix Global SFA Registry analysed long lesions (140–500 mm); of these 42.1% were chronic total occlusions (CTOs).21 Freedom from TLR in long lesions was similar to all lesions (93.0 % long lesions versus 93.6 % all lesions), as well as the 30-day safety (99.3 %). Long lesions (>15 cm in length) were also analysed in the IN.PACT Global Study.9 Of 157 lesions, with a mean length of 26.40 ± 8.61 cm, 60.4 % were CTO. 1-year PP was 91.1 %
(360 days) and 80.7 % (390 days) and 1-year CD TLR was 6.0 % with 40.4 % provisional stenting.22
The IN.PACT Admiral DCB was also evaluated in a real-world registry of 260 patients with 288 lesions longer than 10 cm (mean lesion length of 24 cm).23 The patient population was complex with 65.3 % CTO, 51.7 % de novo lesions, 11.1 % restenosis and 37.2 % ISR. Kaplan-Meier estimated PP was 79.2 ± 2.6 % at 1 year and 53.7 ± 3.4 % at 2 years. Freedom from TLR was 85.4 ± 2.1 % for the entire cohort at 1 year and 68.6 ± 3.0 % at 2 years. In the critical limb ischaemia group only, the amputation rate was 5.3 % at 1 year and 7.9 % at 2 years.23
Table 2 summarises the main characteristics and outcomes of the real-world registries.
Although all DCBs use paclitaxel as the active drug, multiple technical differences exist between each DCB, such as drug dose (ranging from 2 μg/mm2 to 3.5 μg/mm2), formulation (crystalline versus amorphous), excipient type and coating technology.
The ‘correct’ drug dose is under discussion. The drug dose on the balloon surface ranges from 2 μg/mm2 to 3.5 μg/mm2 but the optimal dose is not defined. It is queried whether drug dose should differ according to the patient’s condition (for instance, if they have diabetes or not) or according to the lesion morphologic characteristics (more or less calcific), the type of lesion (stenosis or occlusion) and the lesion length. Moreover, the actual drug dose that is released at the level of the target lesion cannot be precisely evaluated in vivo. This value depends on the lesion location, the time of exposure of the catheter to the blood flow, the lesion morphology, and so on.
However, on the basis of the available results, drug dose seems not to be responsible for the performance of the DCB. Similar rates of PP have been reported for the IN.PACT and the Stellarex DCBs (PP at 2 years; 78.9 % versus 80.3 % for IN.PACT and Stellarex, respectively) despite the use of different drug doses (3.5 μg/mm2 versus 2.0 μg/mm2).10,11,17,18
Gongora et al. evaluated the arterial paclitaxel tissue concentration profiles over time for the IN.PACT Pacific, the Lutonix and the Ranger DCBs.24 Despite the different drug dose, all of the DCBs displayed similar levels of drug tissue concentration following inflation. However, after 24 hours, paclitaxel drug tissue concentration levels decreased to ~20 ng/mg for both IN.PACT and Ranger and to ~5 ng/mg for the Lutonix. At seven days, drug tissue concentration levels for IN.PACT and Ranger decreased to ~50 % and to ~1–2 ng/mg for Lutonix. IN.PACT and Ranger drug tissue concentration levels dropped to ~1–2 ng/mg after 30 days.
DCBs represent an important innovation and established technique in the treatment of atherosclerotic disease of the femoropopliteal region. However, not all the DCBs are the same. Despite the use of the same drug, results are correlated with the technical characteristics of each balloon. Therefore, a ‘no class effect’ has been introduced and each DCB must stand on its own merit. This important concept should be kept in mind when choosing a DCB. We look forwards to further studies providing interesting and important data to confirm the safety and efficacy of this technology and help us to solve the still unanswered questions.






